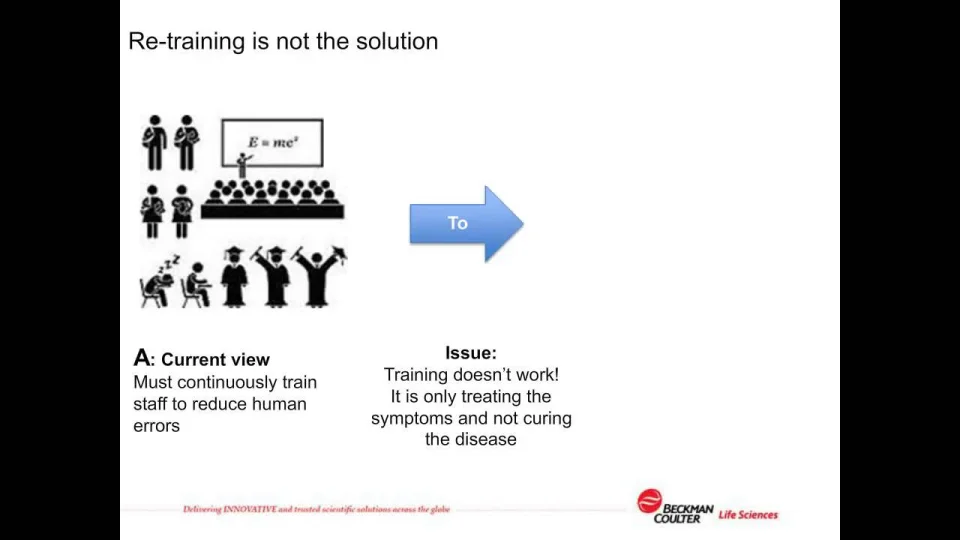
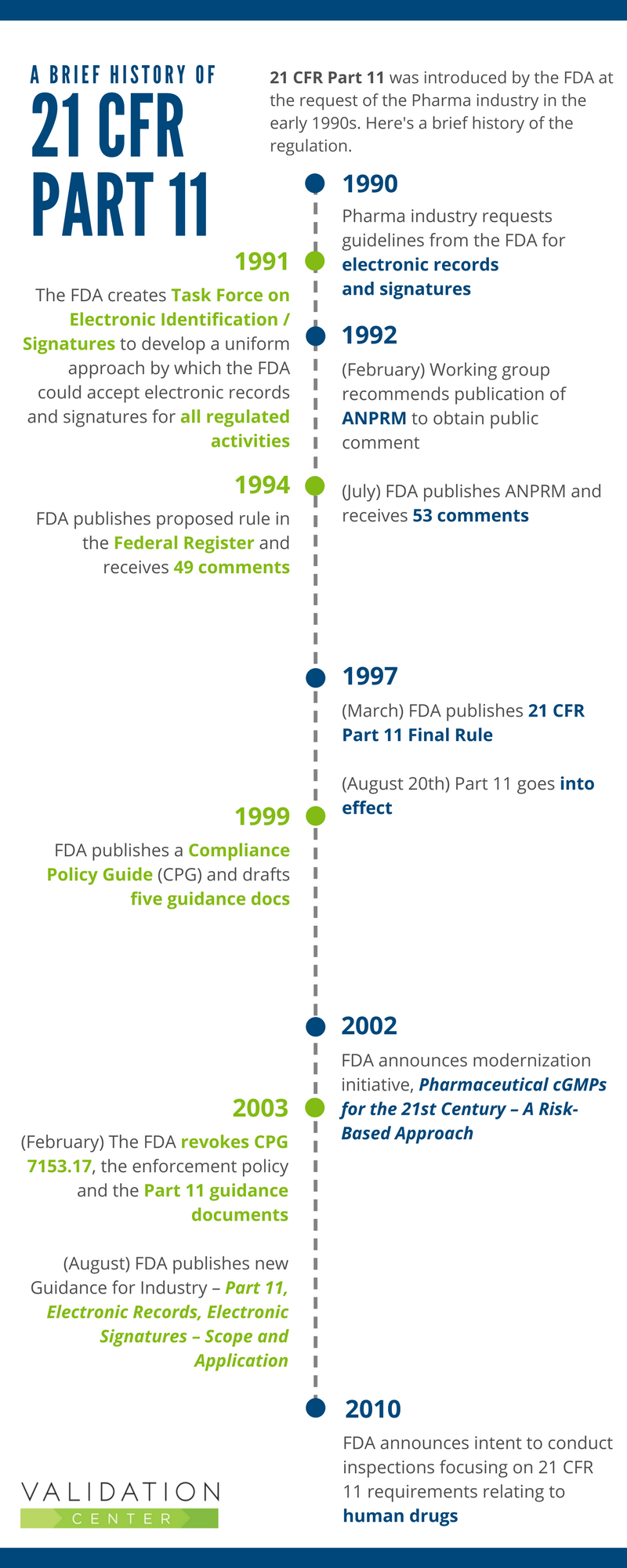
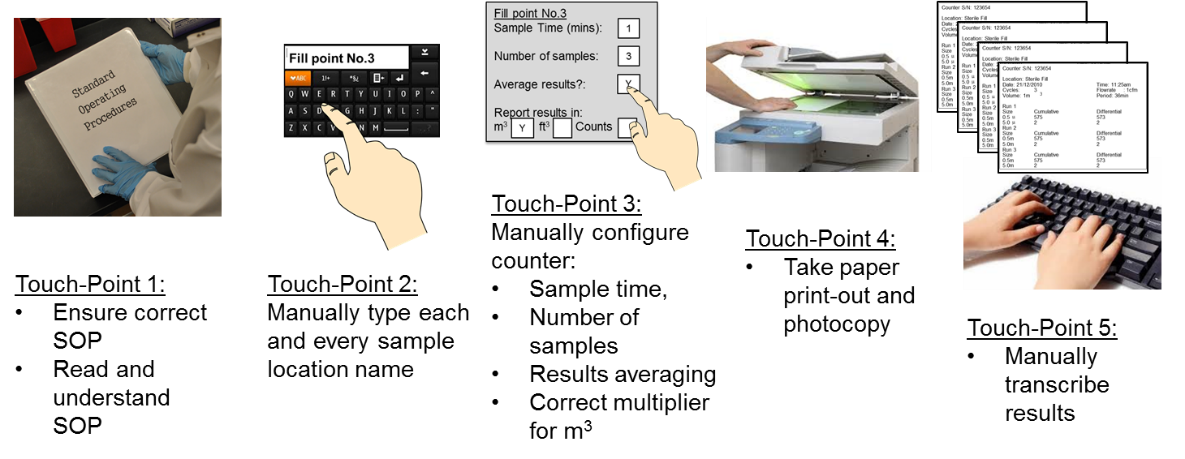
Clarifying and Meeting the Requirements of 21 CFR Part 11 and Data Integrity for Dissolution Testing | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Data Integrity and Privacy – compliance with 21 CFR Part 11, SaaS/Cloud Tickets, Wed, Jan 25, 2023 at 1:00 PM | Eventbrite